Patients Required to Take Part in the GAMBOL Growth Hormone Study at QEHB
July 2023
Sherwin Crisceno at QEHB is looking for patients to take part in the GAMBOL study.
To take part, you must be a patient at QEHB and you must be taking growth hormone replacement therapy.
Sherwin's flyer below gives more information along with his contact details.
July 2023
Sherwin Crisceno at QEHB is looking for patients to take part in the GAMBOL study.
To take part, you must be a patient at QEHB and you must be taking growth hormone replacement therapy.
Sherwin's flyer below gives more information along with his contact details.
___________________________________________________________________________________________
Supply Shortage of Ready Mixed Emergency Hydrocortisone Injection
May 2023
We have been advised that the ready mixed Hydrocortisone Sodium Succinate 100mg from Pan Pharma is out of supply until August 2023.
The Pfizer generic version of Solu-Cortef (Hydrocortisone Sodium Succinate 100mg powder for solution for injection) is widely available with no stock issues. You just need to specify Pfizer in your prescription. This is the powder that you have to mix with sterile water yourself.
May 2023
We have been advised that the ready mixed Hydrocortisone Sodium Succinate 100mg from Pan Pharma is out of supply until August 2023.
The Pfizer generic version of Solu-Cortef (Hydrocortisone Sodium Succinate 100mg powder for solution for injection) is widely available with no stock issues. You just need to specify Pfizer in your prescription. This is the powder that you have to mix with sterile water yourself.
__________________________________________________________________________________________
The Pituitary Foundation's new website
April 2023
The Foundation has recently refreshed its image with a new logo and different colours. The old website was not easy to navigate and many people complained that it was difficult to find the information they were looking for.
The new website was launched today (26th April) and it is well worth having a look at. It looks modern and professional and it is definitely easier to find what you are looking for. The address remains the same pituitary.org.uk
April 2023
The Foundation has recently refreshed its image with a new logo and different colours. The old website was not easy to navigate and many people complained that it was difficult to find the information they were looking for.
The new website was launched today (26th April) and it is well worth having a look at. It looks modern and professional and it is definitely easier to find what you are looking for. The address remains the same pituitary.org.uk
_____________________________________________________________________________________________________________________
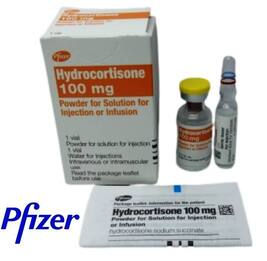
Name change for Solu-Cortef emergency hydrocortisone injection
March 2023
When picking up your next prescription, you might notice a slight packaging difference for your emergency injection medication. The previously branded ‘Solu Cortef’ hydrocortisone injection will now be known as ‘Hydrocortisone 100mg Powder for Solution for Injection or Infusion 1x2ml (vial with diluent)’.
The name change is because the patent on the original has expired and other companies are now able to produce generic versions. Foundation news
March 2023
When picking up your next prescription, you might notice a slight packaging difference for your emergency injection medication. The previously branded ‘Solu Cortef’ hydrocortisone injection will now be known as ‘Hydrocortisone 100mg Powder for Solution for Injection or Infusion 1x2ml (vial with diluent)’.
The name change is because the patent on the original has expired and other companies are now able to produce generic versions. Foundation news
_____________________________________________________________________________________________________________________
Research Opportunity: Experiences of family members of those with pituitary conditions
News Date 06/03/2023
This study is seeking people who are family members of those with pituitary conditions. The study is run by the School of Psychology at University of Plymouth.
This study aims to explore the lived experiences of family members who have a relative with a pituitary condition, to improve our understanding of their experiences of support. The results will be compared with findings from similar studies and will be used to explore how support might be improved in the future.
You will be asked to take part in a one-to-one online interview where you will be asked about your experiences of having a relative with a pituitary condition. You will also be asked about your experiences of the accessibility and availability of services and support. The interview should take no longer than 30-45 minutes. A small donation will be made to the Pituitary Foundation in recognition of your participation.
To take part, or if you would like any further details regarding the study, feel free to contact the principal investigators:
Carly Ellicott: [email protected]
Dr Alyson Norman: [email protected]
News Date 06/03/2023
This study is seeking people who are family members of those with pituitary conditions. The study is run by the School of Psychology at University of Plymouth.
This study aims to explore the lived experiences of family members who have a relative with a pituitary condition, to improve our understanding of their experiences of support. The results will be compared with findings from similar studies and will be used to explore how support might be improved in the future.
You will be asked to take part in a one-to-one online interview where you will be asked about your experiences of having a relative with a pituitary condition. You will also be asked about your experiences of the accessibility and availability of services and support. The interview should take no longer than 30-45 minutes. A small donation will be made to the Pituitary Foundation in recognition of your participation.
To take part, or if you would like any further details regarding the study, feel free to contact the principal investigators:
Carly Ellicott: [email protected]
Dr Alyson Norman: [email protected]
_____________________________________________________________________________________________________________________
Adrenal Infufficiency Research Opportunity
Nov 2022
Researchers at the Universities of Birmingham, Warwick and Kent are at the very beginning of a new project which hopes to develop a prototype sensor that can be worn on the skin to detect the levels of the hormone cortisol.
The long-term goal is that this technology can be tested and developed further to help people who have to take medication to regulate their cortisol levels, like ourselves. Consequently making it much easier to know how much replacement steroid we need throughout the day.
The team are very keen to chat with people who have Addison's disease and adrenal insufficiency early on, to gather our thoughts, ideas and questions about the project – so that pitfalls can be avoided and so that the team understand what people would need most from the technology and their key concerns.
So this is where you come in! Please consider taking part in this pilot project, where through a Zoom consultation event you can help provide lived experience to the research team.
The event will be joined by endocrinologists from Birmingham and Bristol, as well as by people with Cushing’s disease (the other side of the coin in cortisol imbalances). This important event is led by Eder Zavala and Caroline Gillett, who are part of the Centre for Systems Modelling & Quantitative Biomedicine at the University of Birmingham.
Who can take part: People who are under a cortisol replacement drug regime (e.g. primary Addison’s disease and secondary adrenal insufficiency). Are 18 years old or older
What is involved: Take part in an online Zoom discussion lasting approximately 2 hours (with a comfort break in between).
You will be kept up to date with the development of the study as further funding applications are made.
The study is entirely voluntary and participants are free to withdraw at any time.
How to take part: Please e-mail: [email protected] to register your interest and the team will get in touch.
Nov 2022
Researchers at the Universities of Birmingham, Warwick and Kent are at the very beginning of a new project which hopes to develop a prototype sensor that can be worn on the skin to detect the levels of the hormone cortisol.
The long-term goal is that this technology can be tested and developed further to help people who have to take medication to regulate their cortisol levels, like ourselves. Consequently making it much easier to know how much replacement steroid we need throughout the day.
The team are very keen to chat with people who have Addison's disease and adrenal insufficiency early on, to gather our thoughts, ideas and questions about the project – so that pitfalls can be avoided and so that the team understand what people would need most from the technology and their key concerns.
So this is where you come in! Please consider taking part in this pilot project, where through a Zoom consultation event you can help provide lived experience to the research team.
The event will be joined by endocrinologists from Birmingham and Bristol, as well as by people with Cushing’s disease (the other side of the coin in cortisol imbalances). This important event is led by Eder Zavala and Caroline Gillett, who are part of the Centre for Systems Modelling & Quantitative Biomedicine at the University of Birmingham.
Who can take part: People who are under a cortisol replacement drug regime (e.g. primary Addison’s disease and secondary adrenal insufficiency). Are 18 years old or older
What is involved: Take part in an online Zoom discussion lasting approximately 2 hours (with a comfort break in between).
You will be kept up to date with the development of the study as further funding applications are made.
The study is entirely voluntary and participants are free to withdraw at any time.
How to take part: Please e-mail: [email protected] to register your interest and the team will get in touch.
_____________________________________________________________________________________________________________________
Results of The Foundation's 2021 Patient Care Survey
June 2022
The Foundation has published the results of the 2021 patient care survey.
Many pituitary patients shared their experiences and insights into NHS Endocrinology care. This is the largest survey of it’s kind and gives a startling insight into endocrine care in the UK.
You can read the report on The Foundation's website or you can download a pdf copy here.
June 2022
The Foundation has published the results of the 2021 patient care survey.
Many pituitary patients shared their experiences and insights into NHS Endocrinology care. This is the largest survey of it’s kind and gives a startling insight into endocrine care in the UK.
You can read the report on The Foundation's website or you can download a pdf copy here.
_____________________________________________________________________________________________________________________
Society for Endocrinology, Endocrine Nurse Clinical Update - April 2022
Steve and our new CEO, Ren Renwick, manning The Foundation's table at the Hilton Metropole Hotel, NEC
Steve and our new CEO, Ren Renwick, manning The Foundation's table at the Hilton Metropole Hotel, NEC
_____________________________________________________________________________________________________________________

Sponsored Skydive
15 August 2020
On Saturday 15 August 2020, Tanya-Leigh Lane and Emma Hollingsworth did a sponsored skydive to raise money for The Foundation.
Here’s a couple of photos of the daring pair –well done to you both!
If you would like to make a donation, here is the link
15 August 2020
On Saturday 15 August 2020, Tanya-Leigh Lane and Emma Hollingsworth did a sponsored skydive to raise money for The Foundation.
Here’s a couple of photos of the daring pair –well done to you both!
If you would like to make a donation, here is the link
_____________________________________________________________________________________________________________________
New Organ Donation Legislation in England
May 2020
On 20th May 2020, the law in England concerning organ donation consent changed.
The system is now an ‘opt out’ one – this means that all adults over 18 in England will be assumed to have given their consent to be an organ donor when they die.
If you don’t want this automatic donation to apply, it is simple to opt out by recording your decision on the NHS Donor Register. The quickest way to do this is online or you can call the contact centre on 0300 123 2323
Society for Endocrinology/NHS New Steroid Card
March 2020
The Society for Endocrinology and NHS have issued a new adrenal crisis card for steroid-dependent patients. You can download a pdf copy here:
March 2020
The Society for Endocrinology and NHS have issued a new adrenal crisis card for steroid-dependent patients. You can download a pdf copy here:
| new-nhs-emergency-steroid-card.pdf | |
| File Size: | 262 kb |
| File Type: | |
Electronic Prescription Service (EPS)
19 November 2019
NHS will scrap paper prescriptions next month in bid to save £300m over two years
Paper prescriptions will become a thing of a past next month under an NHS plan to save £300million over two years. From November 18th 2019, all GP practices around the country will switch to electronic prescribing. Patients will no longer have to hand in a physical copy of their prescription to chemists - medication details will be sent directly from doctors to a local pharmacy nominated by the patient where it can be collected after the patient gives their name and date of birth.
Patients who want a paper prescription can still request one, but under the new scheme all prescriptions will be printed with a unique barcode rather than a GP signature. This means they can walk into any pharmacy in the country where the barcode can be scanned by a pharmacist to download details of the medication. The Electronic Prescription Service (EPS) also means patients will no longer have to visit their GP to get a repeat prescription as drugs can be prescribed for up to a year using an electronic signature.
Health Minister, Jo Churchill said: ‘Digitising the entire prescription service is a key part of keeping up the drive to make the NHS fit for the 21st century.
'This will free up vital time for GPs and allow pharmacists to spend more time with their patients, and save millions of pounds a year.
‘It’s another important step towards eventually making all prescriptions paperless. We are continuing to improve technology across the NHS which will ultimately improve care for patients.’
Electronic prescriptions were introduced ten years ago and are already used by around 70 per cent of doctors’ surgeries in the UK. The vast majority of GP practices will fully switch to the new system by the end of the year, with the final rollout complete by next summer.
Professor Helen Stokes-Lampard, chairman of the Royal College of GPs, said: ‘Implementing technology in the best interests of patient care by getting the basics right first is a key part of the College’s future vision for general practice.
'Electronic prescribing is an example of technology that works for GPs and our teams, our colleagues in pharmacies and our patients, and we are pleased to see the latest phase of the scheme being rolled out more widely.’
Under the electronic prescribing system, patients can get their medication in two ways.
They can either nominate a pharmacy which will receive the details directly from their GP or they can receive a paper prescription with the digital barcode. The medical information is held on a secure NHS database called NHS Spine and will allow a patient’s prescription to be accessed quickly by GPs and pharmacies.
Dr Ian Lowry, Director of Digital Medicines and Pharmacy at NHS Digital, said: ‘Every prescription that is sent electronically saves money for the NHS by increasing efficiency.
‘The system is also safer and more secure, as prescriptions can’t be lost and clinicians can check their status online. This is a huge milestone to reach, and one which benefits patients, GPs, pharmacists and the NHS as a whole.’
Simon Dukes, Pharmaceutical Services Negotiating Committee Chief Executive, said: ‘We are very pleased to see the Electronic Prescription Service rolling out nationally so that even more patients can take advantage of this digital NHS service.
‘Pharmacies across England have been piloting electronic prescriptions for some time and they will benefit from the simpler and more secure processes that this final rollout will bring. Freeing up GPs’ vital time.’
Daily Mail, 19th October 2019
19 November 2019
NHS will scrap paper prescriptions next month in bid to save £300m over two years
Paper prescriptions will become a thing of a past next month under an NHS plan to save £300million over two years. From November 18th 2019, all GP practices around the country will switch to electronic prescribing. Patients will no longer have to hand in a physical copy of their prescription to chemists - medication details will be sent directly from doctors to a local pharmacy nominated by the patient where it can be collected after the patient gives their name and date of birth.
Patients who want a paper prescription can still request one, but under the new scheme all prescriptions will be printed with a unique barcode rather than a GP signature. This means they can walk into any pharmacy in the country where the barcode can be scanned by a pharmacist to download details of the medication. The Electronic Prescription Service (EPS) also means patients will no longer have to visit their GP to get a repeat prescription as drugs can be prescribed for up to a year using an electronic signature.
Health Minister, Jo Churchill said: ‘Digitising the entire prescription service is a key part of keeping up the drive to make the NHS fit for the 21st century.
'This will free up vital time for GPs and allow pharmacists to spend more time with their patients, and save millions of pounds a year.
‘It’s another important step towards eventually making all prescriptions paperless. We are continuing to improve technology across the NHS which will ultimately improve care for patients.’
Electronic prescriptions were introduced ten years ago and are already used by around 70 per cent of doctors’ surgeries in the UK. The vast majority of GP practices will fully switch to the new system by the end of the year, with the final rollout complete by next summer.
Professor Helen Stokes-Lampard, chairman of the Royal College of GPs, said: ‘Implementing technology in the best interests of patient care by getting the basics right first is a key part of the College’s future vision for general practice.
'Electronic prescribing is an example of technology that works for GPs and our teams, our colleagues in pharmacies and our patients, and we are pleased to see the latest phase of the scheme being rolled out more widely.’
Under the electronic prescribing system, patients can get their medication in two ways.
They can either nominate a pharmacy which will receive the details directly from their GP or they can receive a paper prescription with the digital barcode. The medical information is held on a secure NHS database called NHS Spine and will allow a patient’s prescription to be accessed quickly by GPs and pharmacies.
Dr Ian Lowry, Director of Digital Medicines and Pharmacy at NHS Digital, said: ‘Every prescription that is sent electronically saves money for the NHS by increasing efficiency.
‘The system is also safer and more secure, as prescriptions can’t be lost and clinicians can check their status online. This is a huge milestone to reach, and one which benefits patients, GPs, pharmacists and the NHS as a whole.’
Simon Dukes, Pharmaceutical Services Negotiating Committee Chief Executive, said: ‘We are very pleased to see the Electronic Prescription Service rolling out nationally so that even more patients can take advantage of this digital NHS service.
‘Pharmacies across England have been piloting electronic prescriptions for some time and they will benefit from the simpler and more secure processes that this final rollout will bring. Freeing up GPs’ vital time.’
Daily Mail, 19th October 2019
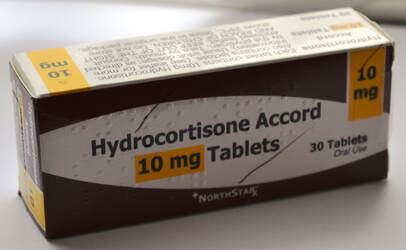
Accord 10mg Hydrocortisone Tablets
09 September 2019
In March (see news item below), Auden McKenzie tablets were rebranded as 'Hydrocortisone Accord'.
Accord Healthcare has now changed the packaging again but the tablets remain exactly the same.
09 September 2019
In March (see news item below), Auden McKenzie tablets were rebranded as 'Hydrocortisone Accord'.
Accord Healthcare has now changed the packaging again but the tablets remain exactly the same.
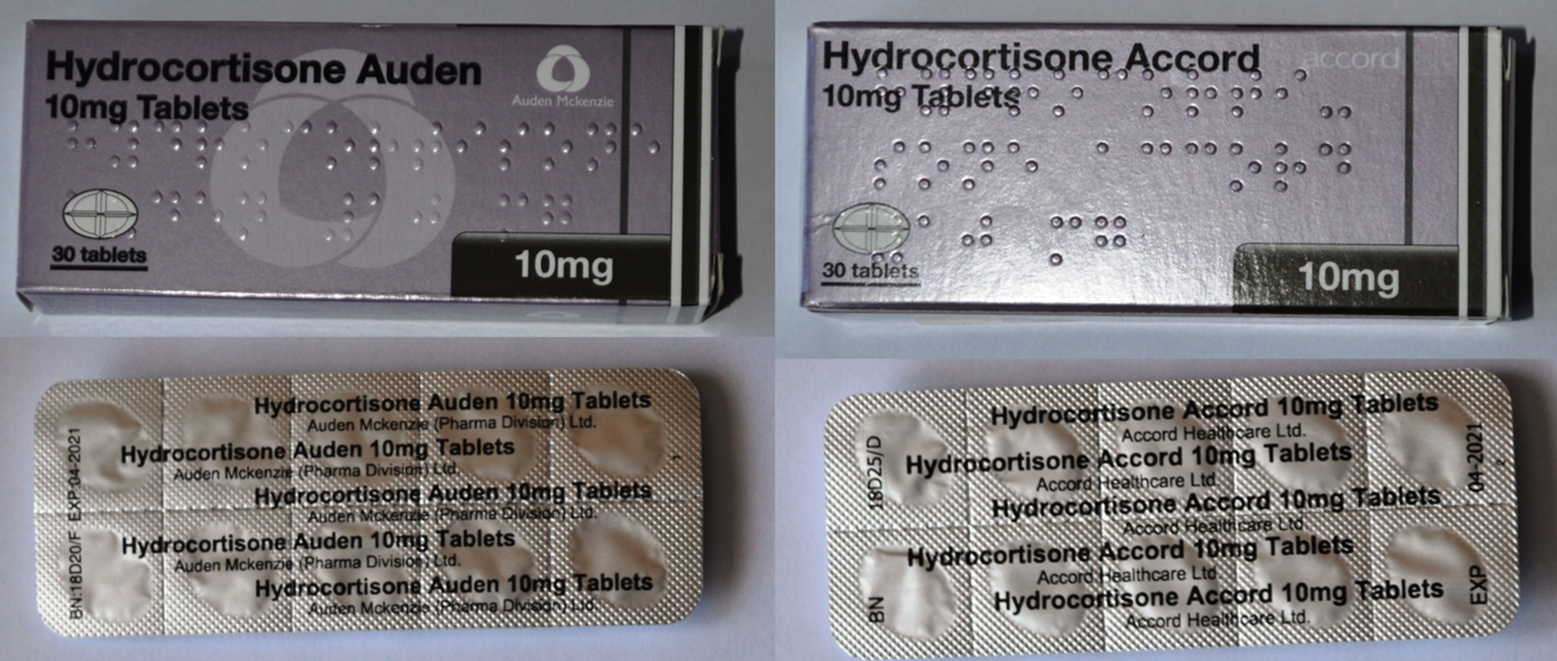
Auden McKenzie 10mg Hydrocortisone Tablets
25 March 2019
Some of you might have noticed that your latest supply of hydrocortisone tablets now has the name of 'Hydrocortisone Accord' instead of 'Hydrocortisone Auden' - don't panic, they are they same tablets!
Auden McKenzie was acquired by Actavis and Actavis has recently rebranded its products as Accord.
The new boxes are almost identical to the old ones except that they are a bit deeper and say 'Accord'. The tablets are exactly the same.
25 March 2019
Some of you might have noticed that your latest supply of hydrocortisone tablets now has the name of 'Hydrocortisone Accord' instead of 'Hydrocortisone Auden' - don't panic, they are they same tablets!
Auden McKenzie was acquired by Actavis and Actavis has recently rebranded its products as Accord.
The new boxes are almost identical to the old ones except that they are a bit deeper and say 'Accord'. The tablets are exactly the same.
Emergency Hydrocortisone Management - QEHB information sheets
January 2018
At The Foundation’s Conference in Birmingham last November, QEHB Lead Endocrine Nurse Sherwin Criseno gave a presentation about replacement hydrocortisone and the management of adrenal crisis.
During his talk, he said that the QEHB endocrine team had recently introduced two documents for patients to show to ambulance and medical staff. These A4 documents are similar to the ones which you can download from The Foundation’s website but the main difference is that the Endocrine Nurses will personalise their documents by adding your hospital patient details sticker to each document.
This makes the documents more ‘official’ because they are on QEHB headed paper and carry your personal details.
The documents are:
Management of adrenal crisis – important information for ambulance and medical staff
Management of glucocorticoid requirement during surgery – important information for surgical, medical and nursing staff
If you would like copies of these two documents, please contact the Endocrine Nurses at the QEHB – it will help if you quote your hospital number
January 2018
At The Foundation’s Conference in Birmingham last November, QEHB Lead Endocrine Nurse Sherwin Criseno gave a presentation about replacement hydrocortisone and the management of adrenal crisis.
During his talk, he said that the QEHB endocrine team had recently introduced two documents for patients to show to ambulance and medical staff. These A4 documents are similar to the ones which you can download from The Foundation’s website but the main difference is that the Endocrine Nurses will personalise their documents by adding your hospital patient details sticker to each document.
This makes the documents more ‘official’ because they are on QEHB headed paper and carry your personal details.
The documents are:
Management of adrenal crisis – important information for ambulance and medical staff
Management of glucocorticoid requirement during surgery – important information for surgical, medical and nursing staff
If you would like copies of these two documents, please contact the Endocrine Nurses at the QEHB – it will help if you quote your hospital number

Wed 10th October 2012
Today, ViroPharma Inc announced the long-awaited launch of Plenadren in England
Plenadren is taken orally as a single dose in the morning and has been designed to provide a dual release pattern of hydrocortisone. The outer coating provides a high concentration of cortisol in the morning, meeting the body’s requirements. The extended-release core provides a smooth cortisol release throughout the day, followed by a cortisol-free interval during the night, avoiding the unphysiological afternoon/evening peaks
observed with standard treatment. Furthermore, Plenadren is proven to be well tolerated with patients reporting favourable impact on quality of life compared to standard treatment, in terms of psychosocial and cognitive functioning, and psychological general well-being. In an 18-month long-term follow-up trial, 98.5% of patients rated tolerability as ‘very well’, ‘well’ or ‘acceptable’.
The Plenadren tablet is available in 5 and 20 mg strengths. The immediate-release outer coating provides physiological cortisol concentrations within 20 minutes of intake and the extended-release core provides a smooth serum cortisol level with reduced exposure in the late afternoon and over the 24 hour period.
“For many years there has been a need for a new oral hydrocortisone replacement therapy in adrenal insufficiency. Plenadren is an innovative once daily dual release hydrocortisone that provides a more physiological plasma cortisol profile avoiding the afternoon/evening peaks associated with conventional replacement therapy that in turn have been associated with adverse metabolic outcomes. The added improvement in quality of life will be a real boost for many patients with primary and secondary adrenal failure” said Professor Paul Stewart, Professor of Medicine and Dean of Medicine Endocrinology, Diabetes and Metabolism at the University of Birmingham.
Today, ViroPharma Inc announced the long-awaited launch of Plenadren in England
Plenadren is taken orally as a single dose in the morning and has been designed to provide a dual release pattern of hydrocortisone. The outer coating provides a high concentration of cortisol in the morning, meeting the body’s requirements. The extended-release core provides a smooth cortisol release throughout the day, followed by a cortisol-free interval during the night, avoiding the unphysiological afternoon/evening peaks
observed with standard treatment. Furthermore, Plenadren is proven to be well tolerated with patients reporting favourable impact on quality of life compared to standard treatment, in terms of psychosocial and cognitive functioning, and psychological general well-being. In an 18-month long-term follow-up trial, 98.5% of patients rated tolerability as ‘very well’, ‘well’ or ‘acceptable’.
The Plenadren tablet is available in 5 and 20 mg strengths. The immediate-release outer coating provides physiological cortisol concentrations within 20 minutes of intake and the extended-release core provides a smooth serum cortisol level with reduced exposure in the late afternoon and over the 24 hour period.
“For many years there has been a need for a new oral hydrocortisone replacement therapy in adrenal insufficiency. Plenadren is an innovative once daily dual release hydrocortisone that provides a more physiological plasma cortisol profile avoiding the afternoon/evening peaks associated with conventional replacement therapy that in turn have been associated with adverse metabolic outcomes. The added improvement in quality of life will be a real boost for many patients with primary and secondary adrenal failure” said Professor Paul Stewart, Professor of Medicine and Dean of Medicine Endocrinology, Diabetes and Metabolism at the University of Birmingham.



